|
At the time of sacrifice (week 8), histological and immunofluorescent analyses were performed. For follow-up, ♜T was performed at weeks 0, 4, and 8 weeks. For in vivo testing, two 2 mm circular defects were created in the frontal and parietal bones of NOD/SCID mice and treated with Ink-Bone, Ink-Bone + BM-MSC-BMP6, Ink-Bone + iMSC-BMP6, Ink-Bone + iNCC-MPC-BMP6, or left untreated. The osteogenic potential of bio-ink-cell constructs were evaluated in osteogenic media or nucleofected with BMP6 using qRT-PCR and in vitro μCT. Cell viability, osteogenic potential of cells, and bio-ink (Ink-Bone or GelXa) combinations were investigated in vitro using bioluminescent imaging. The study aimed to evaluate the potential of iPSC-derived cells via neural crest or mesoderm overexpressing BMP6 and embedded in 3D printable bio-ink to generate viable bone graft alternatives for cranial reconstruction. Overexpression of human recombinant bone morphogenetic protein (BMP)6 efficiently stimulates bone formation. Prior research demonstrated the potential of bone marrow-derived MSCs (BM-MSCs) and iPSC-derived mesenchymal progenitor cells via the neural crest (NCC-MPCs) or mesodermal lineages (iMSCs) to be promising cell source for bone regeneration. Induced pluripotent stem cell (iPSC) differentiation is a powerful tool to generate mesenchymal stromal cells (MSCs). 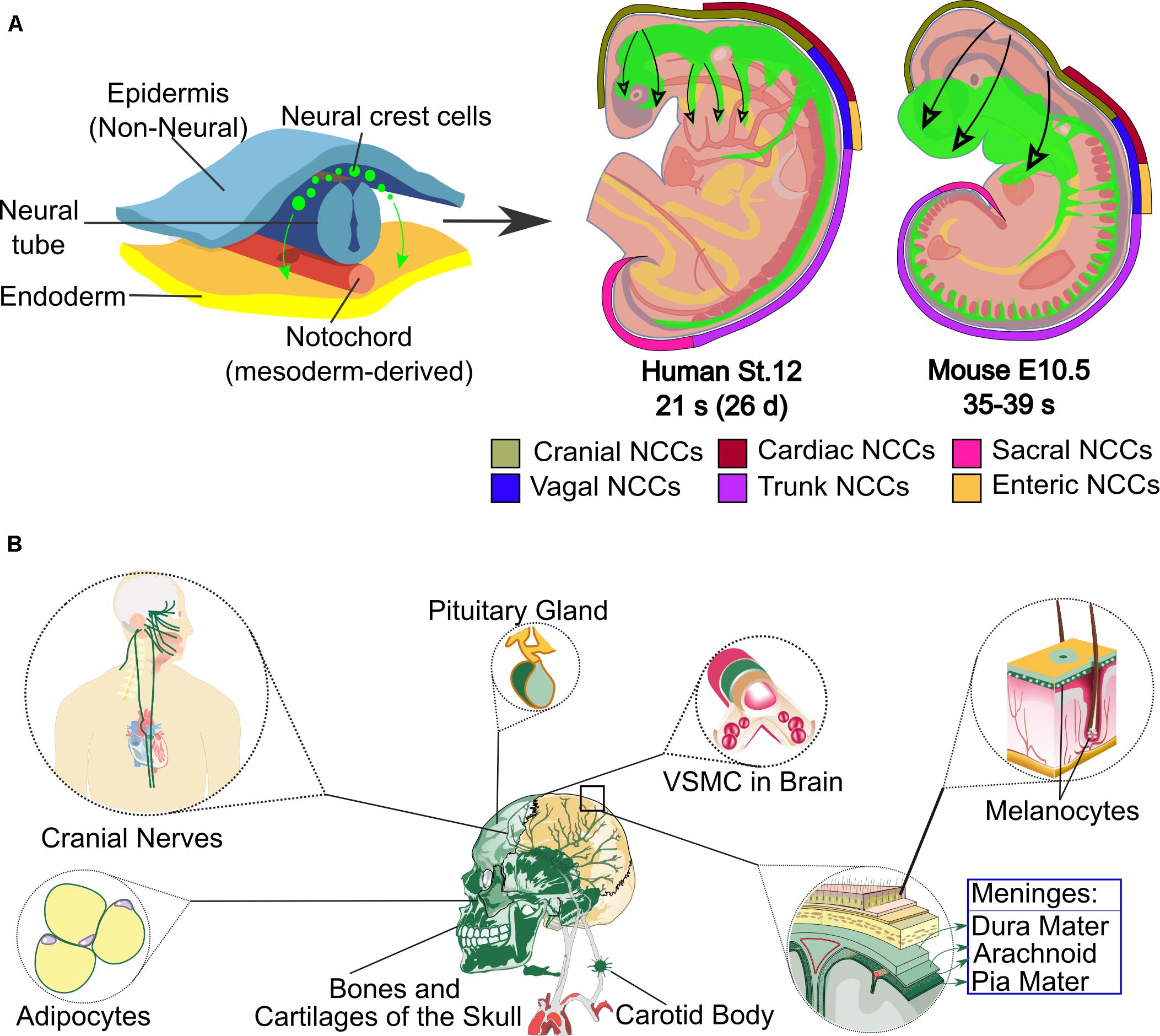
Cranial bone loss presents a major clinical challenge and new regenerative approaches to address craniofacial reconstruction are in great demand.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
